Introduction
Protocols and purification methods constitute indispensable pillars in the fields of molecular biology, biochemistry, and biotechnology. These methodologies are pivotal for ensuring the reproducibility, precision, and preservation of the functional integrity of biomolecules, including proteins, peptides, and antibodies. By facilitating the isolation and characterization of these entities, such approaches underpin advancements in both fundamental research and applied sciences. This overview delineates key concepts and applications, serving as a foundational resource for researchers. For those requiring in-depth procedural guidance and sophisticated techniques, a comprehensive course is recommended to delve into specialized protocols and optimization strategies.
The evolution of protein purification techniques has been driven by the need for high-purity biomolecules essential for structural elucidation, functional assays, and therapeutic development. Advances in chromatography and other separation methods have enabled scalable, efficient processes that minimize contaminants while maximizing yield.
Experimental Protocols
Experimental protocols provide meticulous, step-by-step directives for conducting laboratory investigations, thereby guaranteeing reproducibility and accuracy. These frameworks are crucial for validating biomolecular assays, mitigating variability, and ensuring data integrity across experiments. Protocols typically encompass sample preparation, reaction conditions, controls, and analytical endpoints, tailored to the specific biomolecule under study.
In practice, protocols begin with cell lysis or tissue disruption to release target biomolecules, followed by clarification steps such as centrifugation to remove debris. Standardization of these steps is vital for downstream applications, as inconsistencies can compromise assay reliability.
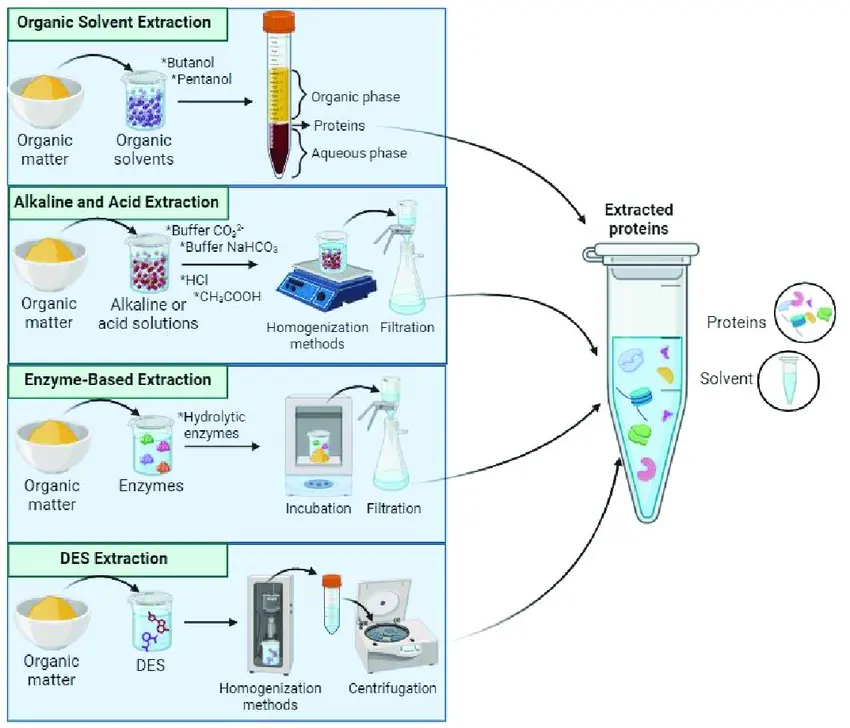
Purification Methods
Purification methods focus on the isolation of proteins, peptides, and antibodies from complex mixtures, employing techniques that exploit physicochemical properties such as size, charge, hydrophobicity, and affinity. Common modalities include chromatography (e.g., affinity, ion-exchange, size-exclusion), electrophoresis, high-performance liquid chromatography (HPLC), and ultrafiltration. These methods are essential for achieving high purity levels required for subsequent research and therapeutic utilizations.
Affinity chromatography, for instance, leverages specific ligand-protein interactions to selectively bind and elute the target molecule. Ion-exchange chromatography separates based on charge differences, while size-exclusion relies on molecular weight. HPLC offers high-resolution separation under pressure, and ultrafiltration uses membrane-based filtration for concentration and desalting. Recent advancements incorporate magnetic separation and aqueous two-phase systems for enhanced efficiency
Applications
The applications of purified biomolecules span diverse domains:
Research: Enabling structural studies via crystallography or NMR, functional assays to probe enzymatic activity, and target validation in drug discovery
Diagnostics: Facilitating biomarker detection through immunoassays and the development of sensitive detection platforms.
Therapeutics: Supporting the production of biologics, recombinant proteins, and peptide-based medecine, with purification ensuring safety and efficacy in clinical settings.
Why This Matters
Comprehension of protocols and purification is imperative for robust experimental design, precise molecular data interpretation, and the attainment of high-quality, reproducible research outcomes. These elements are foundational to translational and clinical applications, where impure or inconsistent biomolecules could lead to erroneous conclusions or therapeutic failures. In an era of personalized medicine and biopharmaceutical innovation, mastery of these techniques enhances efficiency, reduces costs, and complies with regulatory standards.