Counter Current Immunoelectrophoresis Protocol
Introduction
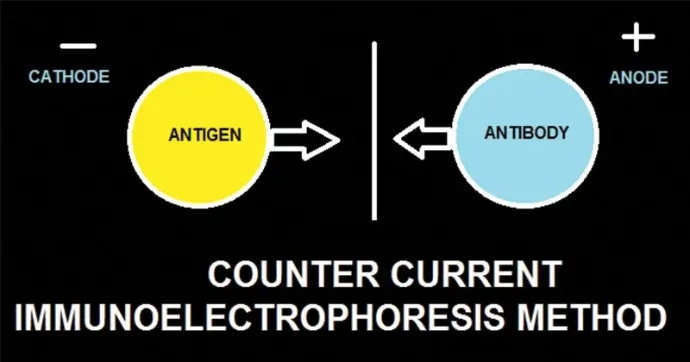
Counter Current Immunoelectrophoresis (CCIE), also known as counterimmunoelectrophoresis (CIE), is a classical immunological technique employed to detect antigen–antibody interactions via electrophoretic migration. This method integrates principles of immunodiffusion with electrophoresis to expedite the formation of visible immune complexes. It is particularly valued in laboratory settings for analyzing antigen presence, assessing antibody specificity, and facilitating experimental immunology workflows. Historically, CCIE has played a pivotal role in immunodiagnostics, offering a rapid, cost-effective alternative to more contemporary assays..
Principle of Counter Current Immunoelectrophoresis
In CCIE, antigens and antibodies are introduced into separate wells within an agarose gel matrix. Upon application of an electric field, the antigen (typically negatively charged at physiological pH) migrates toward the anode, while the antibody (often positively charged or less negatively charged) moves toward the cathode. This counter-directional migration accelerates their encounter, leading to the formation of a visible precipitation line at the equivalence zone where antigen–antibody concentrations are optimal for complexation. This precipitin line indicates specific biomolecular recognition, providing a qualitative detection method with high sensitivity for soluble antigens
Experimental Setup
The technique necessitates a controlled electrophoretic environment and a prepared gel matrix. Essential components include:
- Agarose gel (typically 1% w/v) prepared in a barbital or phosphate buffer (pH 8.2–8.6) to optimize charge-based migration.
- Antigen sample solution (e.g., microbial extracts or serum dilutions).
- Antibody solution (polyclonal or monoclonal antisera).
- Electrophoresis chamber equipped with a power supply (constant voltage of 5–10 V/cm).
- Gel casting plate with pre-punched wells (3–5 mm diameter, spaced 3–5 mm apart) for sample loading.
The gel serves as a semi-solid support medium that permits sieving and controlled diffusion under the electric field, minimizing non-specific interactions.
Basic Protocol Overview
A standardized protocol for CCIE typically encompasses the following steps:
- Gel Preparation: Dissolve agarose in electrophoresis buffer by heating, then pour onto a clean glass slide or plate to a thickness of 1–2 mm. Allow solidification and punch wells using a template.
- Sample Loading: Pipette 5–10 µL of antigen into the cathodic well and an equivalent volume of antibody into the anodic well. Include positive and negative controls for validation.
- Electrophoresis Migration: Connect the chamber to the power supply and apply current for 30–60 minutes at room temperature. Monitor migration to prevent overheating.
- Immune Complex Formation: As molecules converge, insoluble immune complexes precipitate, forming a distinct arc or line visible without staining.
- Result Visualization and Interpretation: Examine the gel under indirect light or with protein stains (e.g., Coomassie Brilliant Blue) if needed. The presence of a precipitin line confirms antigen–antibody interaction; its position and intensity can indicate relative concentrations.
Optimization is crucial, as excessive voltage may distort lines, while suboptimal concentrations could yield false negatives.
Applications in Research
CCIE has been instrumental in various research domains, including:
- Detection of microbial antigens, such as in pneumococcal infections or mycoplasma diagnostics.
- Antibody specificity testing for serological profiling.
- Immunological assay development, particularly in veterinary medicine for diseases like contagious bovine pleuropneumonia or porcine cysticercosis.
- Biomolecular interaction studies in virology, such as dengue antibody quantitation.
Although supplanted by advanced methods like ELISA or Western blotting in many clinical contexts due to higher throughput and quantification capabilities, CCIE persists as an educational tool and in resource-limited settings for rapid screening.
Relationship with Other Electrophoresis Techniques
CCIE belongs to a suite of electrophoretic methods for biomolecular analysis, including rocket immunoelectrophoresis (for antigen quantification) and crossed immunoelectrophoresis (for complex mixture resolution). These techniques exploit charge-dependent migration in electric fields for:
- Protein separation based on size and charge.
- Antigen identification in heterogeneous samples.
- Antibody characterization in serological studies.
- Molecular purity evaluation post-purification.
CCIE's counter-current design distinguishes it by enhancing speed and sensitivity over passive diffusion methods like Ouchterlony double immunodiffusion.
Experimental Considerations
Successful implementation hinges on several parameters:
- Buffer composition (ionic strength affects migration rates).
- Gel concentration (higher agarose restricts larger molecules).
- Antigen and antibody titers (equivalence zone optimization via titration).
- Voltage and duration (typically 60–120 V for 45–90 minutes to avoid joule heating).
Troubleshooting common issues, such as faint lines (due to low concentrations) or artifacts (from impurities), enhances reproducibility. Safety protocols, including handling of biohazards and electrical equipment, are imperative in laboratory practice.
Scientific Context
CCIE significantly advanced early immunodiagnostic frameworks, enabling rapid detection in infectious diseases before enzyme-linked assays became prevalent. Its contributions to fields like pediatric infectious disease diagnostics (e.g., group B streptococcal infections) underscore its historical impact. Today, it serves as a foundational reference in immunology education and methodological training within life science laboratories worldwide
Educational Resources
For readers interested in deeper methodological understanding, a detailed educational document is available.
Counter Current Immunoelectrophoresis and Electrophoresis Protocols.pdf