Isolation and Handling of Primary Mouse Embryonic Fibroblasts: Protocol Overview
Introduction
Primary Mouse Embryonic Fibroblasts (MEFs) serve as a foundational model in molecular and cellular biology, facilitating investigations into gene expression, cell cycle regulation, signal transduction pathways, and reprogramming for stem cell research. These cells, derived from embryonic connective tissue, offer a versatile platform for in vitro studies due to their robustness and amenability to genetic modifications. Ensuring proper isolation and handling is paramount to preserve cell viability, mitigate senescence, and uphold experimental reproducibility, which are critical for downstream applications in both basic and translational research.
Protocol Overview: Isolation of Primary MEFs
The isolation protocol adheres to ethical standards for animal handling and requires sterile techniques to prevent contamination. It encompasses sequential steps to dissociate embryonic tissue into viable fibroblasts.
Embryo Harvesting:
- Euthanize pregnant mice humanely per institutional guidelines (e.g., CO₂ asphyxiation followed by cervical dislocation).
- Dissect the uterine horns aseptically and transfer embryos to ice-cold phosphate-buffered saline (PBS) to preserve tissue integrity.
Tissue Dissection:
- Remove embryonic heads, visceral organs, and hematopoietic tissues (e.g., liver and spleen) under a stereomicroscope to enrich for fibroblastic populations.
- Mince the remaining carcass into 1–2 mm fragments using sterile scalpels.
Enzymatic Digestion:
- Incubate minced tissue in 0.05–0.25% trypsin-EDTA or collagenase solution at 37°C for 10–30 minutes with gentle agitation to achieve dissociation.
- Neutralize enzymes with fetal bovine serum (FBS)-containing medium to halt proteolysis and minimize cell damage.
Cell Collection and Seeding:
- Filter the suspension through a 70–100 μm cell strainer to eliminate undigested debris.
- Centrifuge at 300–500 × g for 5 minutes, resuspend the pellet in growth medium (e.g., DMEM supplemented with 10–15% FBS, 2 mM L-glutamine, and antibiotics), and plate at a density of 1–2 × 10^6 cells per 100 mm dish.
Culture Conditions:
- Incubate at 37°C in a 5% CO₂ humidified atmosphere.
- Replace medium every 2–3 days to remove debris and promote attachment.
This protocol yields MEFs with >90% viability when optimized. For visual reference, a schematic diagram illustrates the key steps in MEF isolation.

Handling and Maintenance
Post-isolation, meticulous maintenance is essential to sustain MEF functionality:
- Passaging: Subculture at 70–80% confluency using trypsin-EDTA to prevent contact inhibition and senescence. Split ratios of 1:3–1:5 are typical.
- Cryopreservation: Freeze cells at passages 1–3 in medium with 10% dimethyl sulfoxide (DMSO) and 20% FBS, storing in liquid nitrogen for long-term viability (>95% recovery upon thawing).
- Avoiding Senescence: Limit experiments to passages 2–5, as higher passages exhibit telomere shortening and reduced proliferation.
- Contamination Control: Employ laminar flow hoods, monitor for mycoplasma via PCR, and use antibiotics judiciously to avoid resistance.
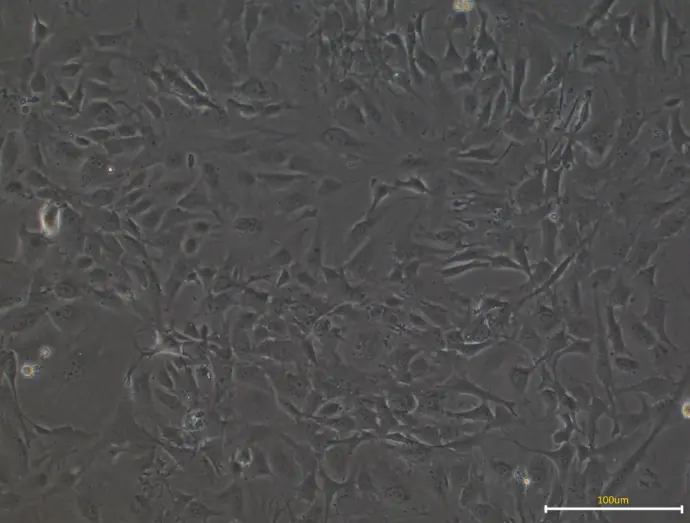
Microscopic examination reveals the spindle-shaped morphology characteristic of healthy MEFs.