DNA Extraction Protocols: Isolation from Blood or Tissue Using Phenol/Chloroform
Introduction
DNA extraction is a foundational technique in molecular biology, enabling the isolation of high-quality nucleic acids essential for a myriad of downstream applications. These include polymerase chain reaction (PCR) and quantitative PCR (qPCR), genomic sequencing, cloning and gene expression analysis, as well as diagnostic assays and biomarker discovery. Despite the advent of commercial kits, the phenol/chloroform extraction method endures as a gold-standard approach for procuring pure, high-molecular-weight DNA from complex biological matrices such as blood and tissue samples. This protocol leverages organic solvent partitioning to achieve superior purity, particularly in scenarios demanding intact genomic DNA for sensitive analyses. The following sections delineate the principles, procedural workflow, and best practices for its implementation in laboratory settings, supported by empirical evidence from peer-reviewed literature.

Biological Background
Deoxyribonucleic acid (DNA) encapsulates the genetic blueprint of all living organisms, comprising a double-helical structure of nucleotide polymers that encode hereditary information. The imperative for extracting intact, contaminant-free DNA stems from its pivotal role in molecular biology research and clinical investigations. Critical considerations in DNA extraction encompass the sample type whether whole blood preserved in anticoagulants like EDTA or fresh/frozen tissue biopsies mitigation of enzymatic degradation by endogenous DNases, minimization of proteinaceous and lipid contaminants, and preservation of DNA structural integrity to facilitate downstream applications such as next-generation sequencing or epigenetic studies.
Empirical studies underscore the efficacy of phenol/chloroform in yielding high-purity DNA from blood and tissue, with yields often exceeding those of salting-out methods while maintaining optimal A260/A280 ratios indicative of minimal protein contamination. For instance, comparative analyses reveal that this method is particularly advantageous for human blood samples, producing DNA suitable for genotyping and long-term biobanking.
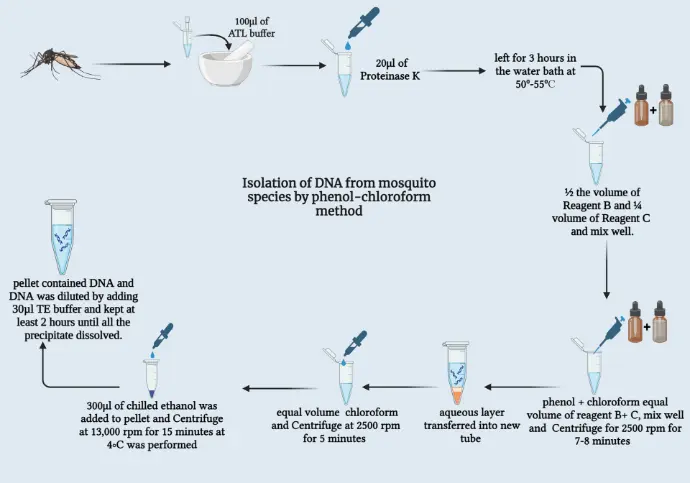
Principle of Phenol/Chloroform Extraction
The phenol/chloroform extraction operates on the principle of liquid-liquid partitioning, wherein nucleic acids are selectively segregated from proteins, lipids, and cellular debris. Phenol, a chaotropic agent, denatures proteins by disrupting hydrophobic interactions, while chloroform enhances phase separation and sequesters residual proteins and lipids into the organic phase. Isoamyl alcohol, often incorporated in a 25:24:1 ratio, mitigates foaming and stabilizes the interphase. Consequently, the hydrophilic DNA partitions into the upper aqueous phase, free from organic contaminants. Subsequent precipitation with alcohols like ethanol or isopropanol concentrates the DNA, yielding a pellet amenable to resuspension.
This methodology affords high-purity DNA, as evidenced by spectrophotometric assessments, rendering it ideal for applications requiring low impurity thresholds, such as metagenomic profiling of microbial communities in wound tissues. Variations, including bead-beating enhancements, further optimize yield from recalcitrant samples like frozen tissues
for more details read more dna_extractios_etc (1).pdf